BIOREMIA objectives
BIOREMIA Network’s mission is to train 15 Early Stage Researchers (ESRs) / PhD students and provide them with the right combination of research-related and transferable competences, empowering them to become the next generation of European experts in biomaterials and medical devices development with reduced infection risks.
BIOREMIA main goals
- To design and produce novel knowledge-based biofilm-resistant materials and surfaces outperforming present competitive solutions.
- To test, measure, simulate, and finally understand the macroscopic response of novel materials and surfaces under work conditions (bio-mechanical and -chemical behavior) and to select the most promising material candidates and processing regimes for up-scaling feasibility studies.
- To train and educate a new generation of innovative young researchers to deal with a cross-disciplinary approach, to understand the needs and inter-dependence of the entire fabrication, processing and characterisation / testing chain, and to apply their knowledge in an industrial practice.
BIOREMIA is structured in 8 work-packages (WPs): the first four are dedicated to research activities, including 15 individual ESR projects, and the remaining four are related to training (WP 5), dissemination (WP 6), management & IPR (WP 7) and ethical issues (WP 8).
Research
Young researchers will have the unique opportunity to be part of a multidisciplinary and inter-sectoral research training programme within two main research areas:
- Bioactive materials with intrinsic antibacterial properties, and
- Nanostructured bactericidal surfaces.
Early Stage Researchers recruited by BIOREMIA will have active involvement in all stages of the biomaterials development process and their application in orthopaedic and dental implants. Our ESRs will gain international and intersectoral experience through cross institute and industrial secondments in order to enhance their employability both inside and outside academia.
WP 1: Material design & Innovative processing
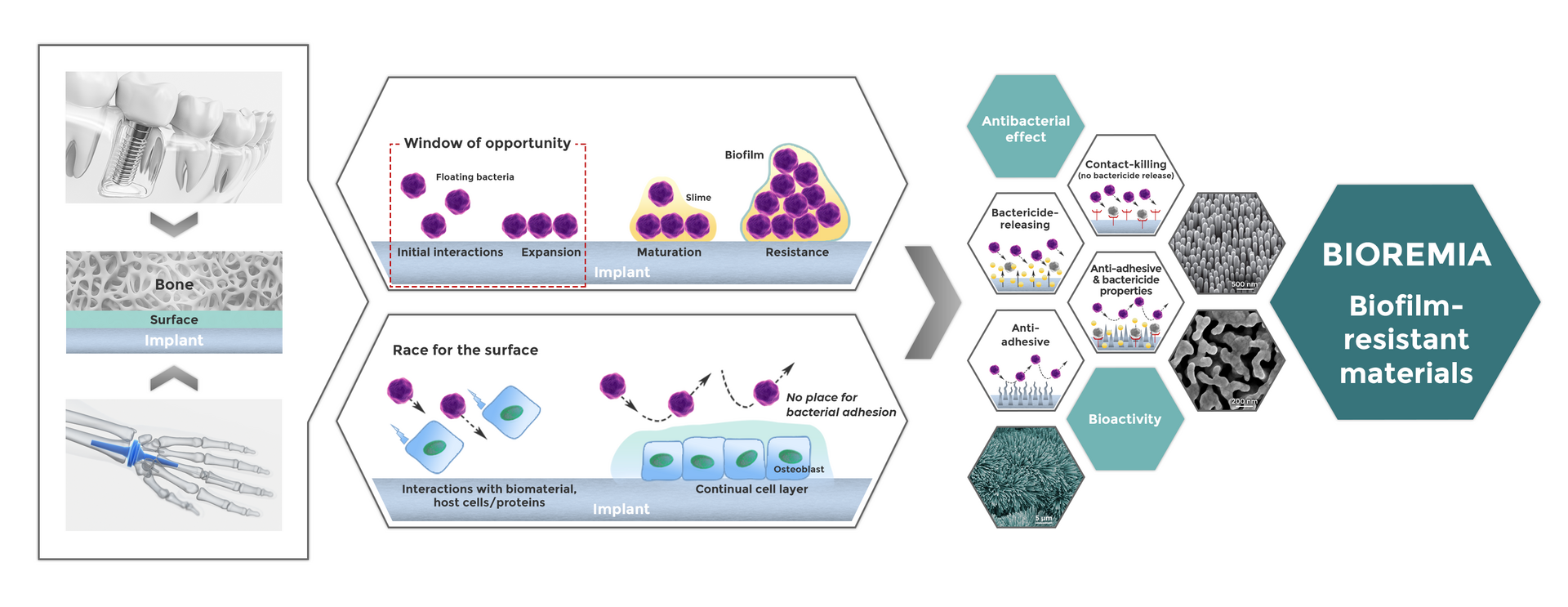
The main objective of WP 1 is to design and produce various biomaterials and coatings with antibacterial and antibiofilm properties, using innovative material processing strategies and surface modification technologies. It contains both the theoretical and experimental work.
BIOREMIA materials selection and design strategy follows three distinctly different mechanisms of antibacterial action:
- Materials / surfaces containing bactericidal elements that kill bacteria in the surrounding areas (‘antibacterial functionality’), resulting in either:
a) release of the bactericidal agent, or
b) in contact-killing without release of the antibacterial agent; the bactericidal agent can be incorporated into the material or coated; - Materials with anti-adhesive properties that are repellent to bacteria, so they cannot attach (‘antifouling functionality’); anti-adhesive properties are obtained by chemical and physical surface modifications;
- Materials combining both functionalities (simultaneously antibacterial and antifouling).
WP 2: In-depth characterisation of materials
This work-package will allow understanding the influence of length scales and complexity of chemical and physical phenomena associated with the targeted functionalities of materials and surfaces. It will provide a full characterisation, by means of the combined infrastructure of the consortium, of the materials and surfaces synthesized in WP1.
The ESRs will screen different material formulations and constructs (bulk, coatings, films) by studying their structure, as well as the physical, chemical and mechanical properties. The most promising materials identified will be in vitro tested in WP 3.
WP 3: Biocompatibility & Bacterial testing
The main objective is to test, measure, simulate, and finally understand the in vitro biological response and antimicrobial / antibiofilm mode of actions of the novel materials and surfaces developed in WP1.
A robust in vitro model based on direct and indirect co-culture enabling a greater understanding of the communication between the different cell types will be established. Using various chemical and biological techniques, the mechanisms by which certain metals may poison the microbial cells will be studied. Specific in vitro bacterial and biofilm tests of the selected specimens will be carried out to demonstrate the advantages of materials and surfaces developed in BIOREMIA when operating as bone-related implants / medical devices. Bacterial strains of clinical relevance for orthopedic and dental implants will be tested, e.g. Staphylococcus (S) aureus, S. epidermidis, P. aeruginosa, Streptococcus oralis.
WP 4: Gateway to industry
In this WP the ESRs will study the feasibility of applying the novel materials and processing methods developed in WP 1 for the production of orthopedic and dental implants / devices. Successful material candidates and laboratory manufacturing / processing routes will be up-scaled; demonstrators will be fabricated. The most important aspects for the manufacturing and surface modification of studied dental and orthopedic medical devices will be assessed and guidelines for optimised processing will be elaborated.
WP 5: Training
This WP will coordinate the training activities, secondments, and the network events in order to deliver the right combination of skills for ESRs. All the fellows have planned secondments in collaboration with our partner organizations and beneficiaries.
WP 6: Dissemination & Outreach
Definition and implementation of a strategy for the dissemination and the promotion of the objectives and results of BIOREMIA to the research community, relevant industry and public stakeholders, schools, wider public and press.
WP 7: Network management & IPR
The objective of WP7 is to ensure a transparent and effective management of scientific, training, financial and administrative issues. It will monitor the progress of the project in concordance with the deliverables and milestones as well as oversee the IP protection.
WP 8: Ethics requirements
The overall objective of this work package is to ensure compliance with the 'ethics requirements' described in the Ethics Review Report so that all the ethical aspects of the project are fulfilled.